The checkpoints of the cell cycle are several points in the eukaryotic Eukaryotes are organisms whose cells have a nucleus enclosed within membranes, unlike prokaryotes, which have no membrane-bound organelles. Eukaryotes belong to the domain Eukaryota or Eukarya. Their name comes from the Greek εὖ and κάρυον. Eukaryotic cells also conta…Eukaryote
What are cell cycle checkpoints?
Cell cycle checkpoints are critical to prevent the cell from progressing to the next phase of the cell cycle before the prior phase has been completed.
What happens when a cell passes the G1 checkpoint?
That is, barring unexpected problems, such as DNA damage or replication errors, a cell that passes the G checkpoint will continue the rest of the way through the cell cycle and produce two daughter cells. The G1 checkpoint. The G1 checkpoint is located at the end of G1 phase, before the transition to S phase.
What are the three checkpoints of the anaphylaxis cycle?
There are a number of checkpoints, but the three most important ones are: The G checkpoint, at the G /S transition. The G checkpoint, at the G /M transition. The spindle checkpoint, at the transition from metaphase to anaphase.
What are the activities of the cell cycle determined by?
The various activities of the cell cycle, then, are determined by the combination of cyclins and CDKs that are active at each stage, as shown in the following table: Can react to outside signals such as growth factors or mitogens. Regulate G2/M checkpoint.
What are the checkpoints in a cell cycle?
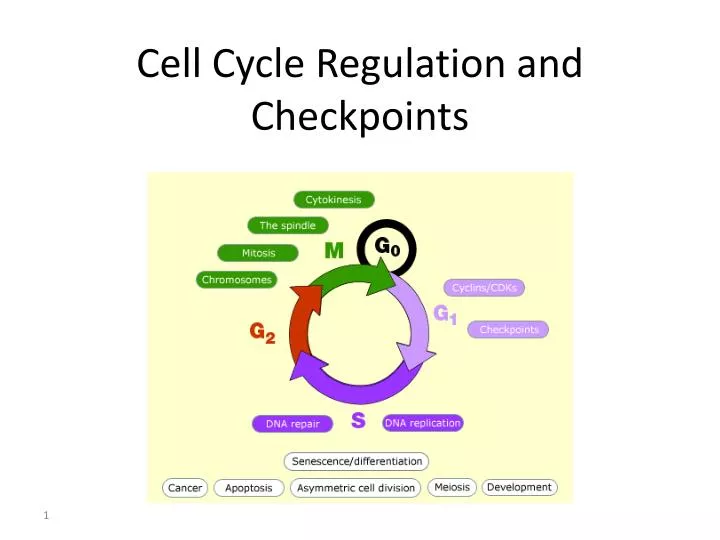
There exist three major cell-cycle checkpoints; the G1/S checkpoint, the G2/M checkpoint, and the spindle assembly checkpoint (SAC).
What is a cell cycle checkpoint quizlet?
What are cell checkpoints? A checkpoint is one of several points in the eukaryotic cell cycle at which the progression of a cell to the next stage in the cycle can be halted until conditions are favorable. These checkpoints occur near the end of G1, at the G2/M transition, and during metaphase.
What is the purpose of checkpoints?
The primary purpose of checkpoints is to deter impaired driving, not to increase arrests. Police generally arrest impaired drivers detected at checkpoints and publicize those arrests, but arrests at checkpoints should not be used as a measure of checkpoint effectiveness.
Why should there be checkpoints in the cell cycle?
Cell cycle checkpoints are critical to prevent the cell from progressing to the next phase of the cell cycle before the prior phase has been completed. Premature entry into the next phase of the cell cycle can result in catastrophic consequences for the cell and cell death.
What are checkpoints list some of the important checkpoints in the cell cycle quizlet?
Terms in this set (3)1st checkpoint: Near end of G1 phase. Cell growth checkpoint, determines whether the cell will divide, if conditions are favorable proteins stimulate a cell to begin the synthesis phase.2nd checkpoint: At the end of G2 phase. ... 3rd checkpoint: At the end of mitosis phase.
What does checkpoint mean in biology?
A checkpoint is a stage in the eukaryotic cell cycle at which the cell examines internal and external cues and "decides" whether or not to move forward with division.
What is the advantage of checkpoint?
A Database Storage Checkpoint keeps track of block change information and thereby enables incremental database backup at the block level. A Database Storage Checkpoint helps recover data from incorrectly modified files.
How is the checkpoint system in the cell cycle helpful in making sure that a cell divides normally?
The cell cycle checkpoints play an important role in the control system by sensing defects that occur during essential processes such as DNA replication or chromosome segregation, and inducing a cell cycle arrest in response until the defects are repaired.
What is the cell cycle control system and how do checkpoints play into this?
A checkpoint is one of several points in the eukaryotic cell cycle at which the progression of a cell to the next stage in the cycle can be halted until conditions are favorable. These checkpoints occur near the end of G1, at the G2/M transition, and during metaphase (Figure 1).
What statements describe how a cell may respond at a checkpoint?
Select all the statements below that describe how a cell may respond at a checkpoint. The cell can halt the cell cycle and enter a permanently non-dividing state. The cell cycle can pause so any damage can be repaired. The cell may initiate the next phase in the cell cycle.
Which of the following are the most important cell cycle checkpoints?
There are three major checkpoints in the cell cycle: one near the end of G1, a second at the G2/M transition, and the third during metaphase. Positive regulator molecules allow the cell cycle to advance to the next stage.
What are the three checkpoints in the cell cycle?
There exist three major cell-cycle checkpoints; the G1/S checkpoint, the G2/M checkpoint, and the spindle assembly checkpoint (SAC).
Why is it important to have a cell cycle checkpoint?
Cell cycle checkpoints are critical to prevent the cell from progressing to the next phase of the cell cycle before the prior phase has been completed. Premature entry into the next phase of the cell cycle can result in catastrophic consequences for the cell and cell death. p53 is a key regulator of the cell cycle and plays a critical role in ...
What are the cell cycle checkpoints in melanoma?
Cell-cycle checkpoints in melanoma are regulated by the p53 and retinoblastoma-associated protein (RB) pathways through p14 and p16 protein ( Box, Vukmer, & Terzian, 2014 ), encoded by the CDKN2A gene ( Schadendorf et al., 2015 ). By preventing the interaction between cyclin D1 and cyclin-dependent kinases 4 and 6 (CDK4/6), and subsequent phosphorylation of RB and activation of general E2F transcription factors, p16 maintains cell-cycle control and functions ( Dimova & Dyson, 2005; Schadendorf et al., 2015 ). In parallel, p14 protein, codified by the same CDKN2A gene, prevents ubiquitylation of p53, mediated by the E3 ubiquitin-protein ligase MDM2; in this way, p14 impedes degradation of p53 and allows expression of p21, whose action converges with that of p16 to inhibit formation of cyclin/CDK complexes and maintain accurate cell cycle progression ( Bieging, Mello, & Attardi, 2014 ). Both p16 and p21 are potent inducers of senescence in melanocytes, the principal mechanism that protects from oncogenic activation of NRAS or BRAF ( Sharpless & Sherr, 2015 ). At the same time, p53 exerts a fundamental function in protecting melanocytes from UV-induced DNA damage, via activation of the proapoptotic factor Bax ( Jhappan, Noonan, & Merlino, 2003 ). Dimerization of the B-cell lymphoma gene-2 protein (Bcl2) with Bax impedes p53-mediated apoptosis, and the ratio of Bcl2 and Bax determines the balance between apoptosis and survival ( Cotter, 2009 ). Bcl2 is produced at high levels in melanocytes, which may explain the atypical resistance to p53-induced apoptosis of this cell type ( Jhappan et al., 2003 ), and the elevated presence of Bcl2 in metastatic melanomas ( Eberle & Hossini, 2008 ).
What is the function of protein phosphorylation in the cell cycle?
Protein phosphorylation and other posttranslational transactions are exploited in checkpoint regulation. Protein phosphorylation promotes cell cycle arrest for all studied checkpoints. Currently, Glc7 dephosphorylation has been implicated in reversing two mitotic cell cycle checkpoints: the spindle checkpoint and DNA damage checkpoint; and one meiotic checkpoint.
Which family phosphatases are involved in cell cycle regulation?
Further, a faster, transcription-independent route for cell cycle regulation exists, via the CDC25 family phosphatases (comprising CDC25A, CDC25B, and CDC25C), which can remove the inhibitory phosphorylation from cyclin-dependent kinases (CDKs).
Is BCL2 produced in melanocytes?
Bcl2 is produced at high levels in melanocytes, which may explain the atypical resistance to p53-induced apoptosis of this cell type ( Jhappan et al., 2003 ), and the elevated presence of Bcl2 in metastatic melanomas ( Eberle & Hossini, 2008 ).
Can HRD cells be affected by checkpoint inhibitors?
Cells that are HRD may be particularly vulnerable to checkpoint inhibitors that prevent the cell cycle checkpoints from appropriately inducing cell arrest, especially as their ability to repair DNA damage is already impaired. For example, one study has demonstrated that Fanconi anemia–deficient tumor cells are sensitive to CHK1 inhibition;
How do cyclins regulate the cell cycle?
Cyclin/CDK complexes regulate the cell cycle both by promoting activites for their respective stages, and by inhibiting activites for future cell cycle stages that must not yet be reached. Therefore cyclins must be able to be both generated and degraded in order for the cell cycle to proceed.
What is the role of APC/C in mitosis?
APC/C is composed of several proteins including but not limited to cullin (APC2), Ring (APC11) and SCF-like protein. APC/C’s role is to ubiquitinate (add ubiquitin tags to) proteins, thus flagging them for degradation by the proteasome.
What is a fully active CDK?
Therefore a fully active CDK is one which is both phosphorylated at the T on the T loop and is bound to a cyclin. The various activities of the cell cycle, then, are determined by the combination of cyclins and CDKs that are active at each stage, as shown in the following table: cell cycle stage. cyclins. CDKs. comments.
What is the binding cleft of CDKs?
They have an ATP binding cleft whose ability to bind ATP is regulated by two mechanisms. First, CDKs have a ‘flexible T loop’ which contains a threonine (T) residue which normally blocks the ATP binding cleft, but not when the T is phosphorylated. Second, cyclins bind CDKs and induce a conformational change that also helps to expose ...
Where is Tem1 associated with?
In yeast, Tem1 becomes associated with the spindle pole body at the centrosome. ‘DNA damage checkpoints’ detect whether DNA has been damaged (e.g. by UV light, chemicals, etc.). The cell cycle arrests in G1 or S, preventing the copying of damaged bases until they can be repaired.
Do CDKs need ATP?
The levels of these proteins remain pretty constant throughout the cell cycle, yet their levels of activity rise and fall cyclically. CDKs need to hydrolize ATP for energy in order to perform phosphorylation. They have an ATP binding cleft whose ability to bind ATP is regulated by two mechanisms.
Popular Posts:
- 1. which institute is best for medical transcription course in bangalore
- 2. what is financial management course
- 3. what is the best lsat prep course to take
- 4. how to write a college course curriculum
- 5. how long is billing and coding course
- 6. how long is medical assistant course
- 7. what is pmp training course
- 8. what is a course management system
- 9. what is business management course
- 10. how long is hvac course